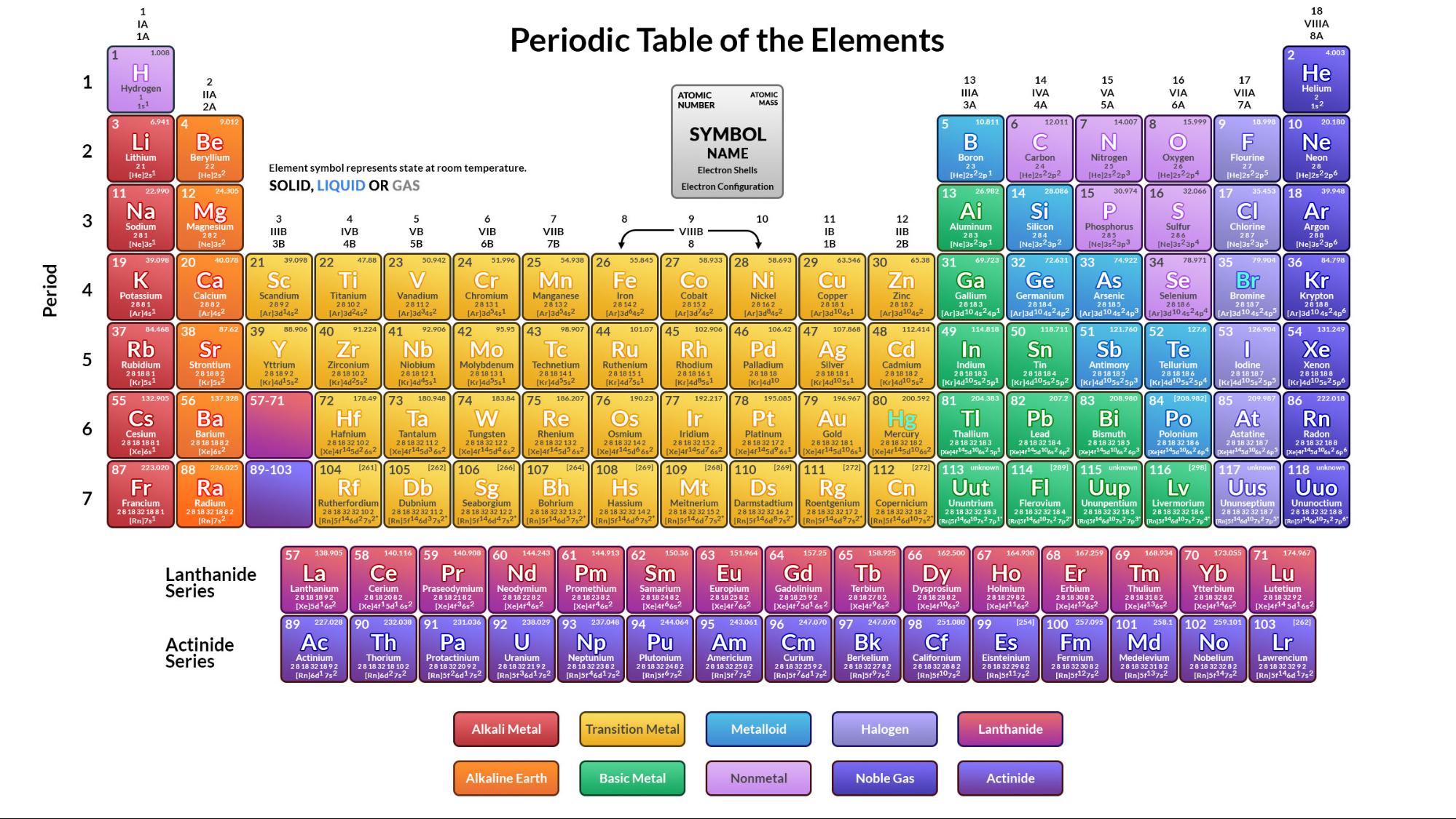
In some cases, he had even predicted how the elements would be discovered, and this was borne out. In his lifetime, Mendeleev had the pleasure of knowing that three of the elements whose existence he had predicted were discovered (gallium, scandium and germanium), and that the physical and chemical properties that he had predicted for these elements and some of their compounds were correct. Legend has it that he saw his classification in a dream. Mendeleev went for a nap on waking up, he wrote out the classification in one go, as it was published in his first communication. He finally copied the arrangement on to a sheet of paper on which can be seen many modifications. Then for several hours he tried to arrange the cards. From this list, he compiled separate cards of the 63 known elements, with their atomic weight and their main chemical properties. Inostrantsev, who later remembered having found Mendeleev in a thoughtful mood, explaining that he had an idea for classification in his head but that he could not find a way of putting it on paper.Īfter this visit, Mendeleev wrote a list of the exact atomic weights in the margin of his book "Principles of Chemistry". Then he received a visit from his friend A. An atom is composed of sub-atomic particles and these cannot be made or destroyed. An atom is defined as the smallest unit that retains the properties of an element. In a second paper dated that same day, Mendeleev made two other unsuccessful attempts at a classification. The smallest particle of an element, which may or may not have an independent existence but always takes place in a chemical reaction is called an atom. So he went off to inspect the dairy, putting off his classifying problem till later on. Mendeleev was trying to arrange the table of elements, but without success. Mendeleev probably received them at breakfast, as suggested by a cup stain on one of them. This day has been retraced in detail and we know that on the morning of 17 February 1869 (1 March in our calendar), Mendeleev received two letters from A. The first version of the periodic table, the one published in 1869, was written in a single day. Periodic Law led to the development of the modern periodic table. Every chemist makes use of Periodic Law, whether consciously or not, when dealing with the chemical elements, their properties, and their chemical reactions. Mendeleev's discovery of the periodic table Periodic Law is considered to be one of the most important concepts in chemistry. The true metals consisting of the alkaline and alkaline earth metals.We also find several families of elements: The reason why it is called the periodic table is that elements with similar properties occur in it at regular intervals, arranged in columns.Įlements in the same column have similar chemical properties. This table contains all the known chemical elements, arranged in order of the number of protons: their atomic number. The Russian chemist Dmitri Mendeleev published his periodic table of the elements in 1869.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
